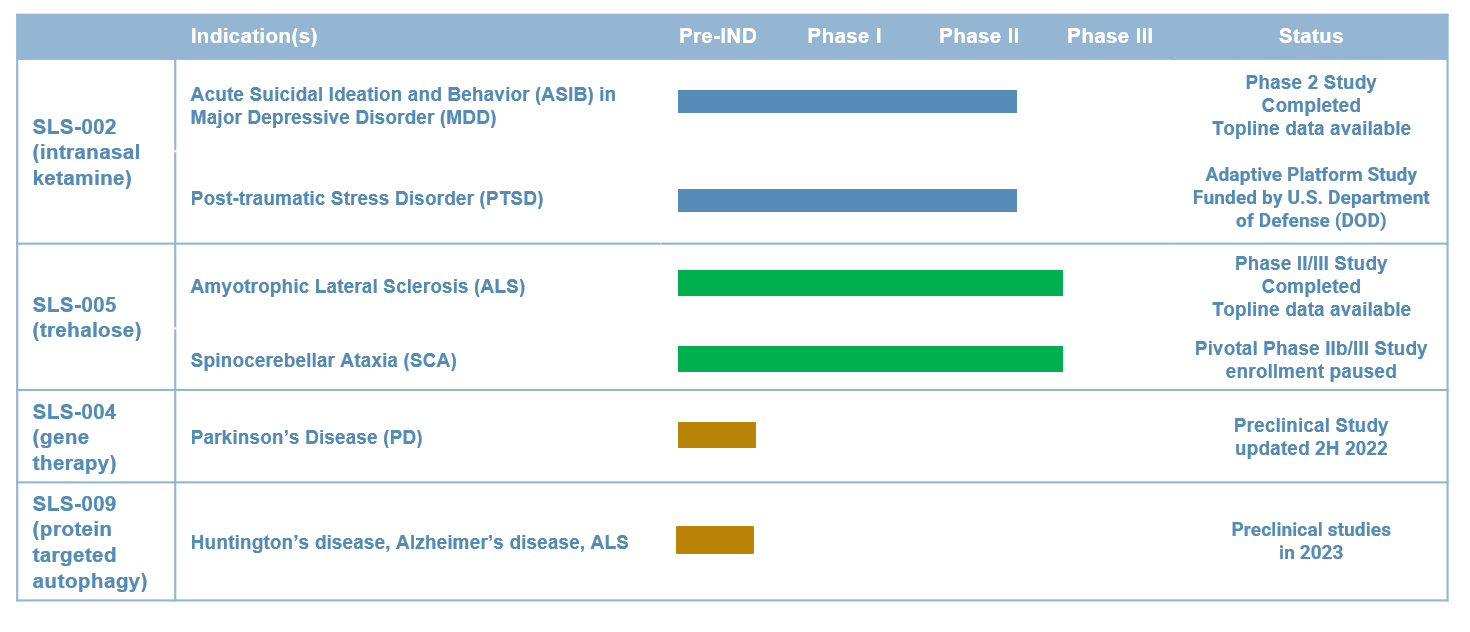
Our Pipeline
Please review our Privacy Policy, Terms of Use, and Accessibility information located at the bottom of this page.

Please review our Privacy Policy, Terms of Use, and Accessibility information located at the bottom of this page.
Latest Posts and Media
Offices
Seelos Therapeutics, Inc.
300 Park Avenue
New York, NY 10022
Incorporated
Nevada
Entity type
Corporation
Identifiers
CIK:1017491
IRS:870449967
© 2026 Seelos Therapeutics, Inc.
| Cookie | Duration | Description |
|---|---|---|
| cookielawinfo-checkbox-analytics | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Analytics". |
| cookielawinfo-checkbox-functional | 11 months | The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". |
| cookielawinfo-checkbox-necessary | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookies is used to store the user consent for the cookies in the category "Necessary". |
| cookielawinfo-checkbox-others | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Other. |
| cookielawinfo-checkbox-performance | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Performance". |
| viewed_cookie_policy | 11 months | The cookie is set by the GDPR Cookie Consent plugin and is used to store whether or not user has consented to the use of cookies. It does not store any personal data. |