About SLS-002 (intranasal ketamine)
SLS-002 (intranasal ketamine) for Acute Suicidal Ideation and Behavior (ASIB) in Major Depressive Disorder (MDD) and Post-Traumatic Stress Disorder (PTSD)
Seelos Therapeutics owns rights to Javelin’s post-operative pain program
- Seven Phase I studies (226 subjects)
- 16 clinical studies (~500 subjects)
SLS-002 Study Design
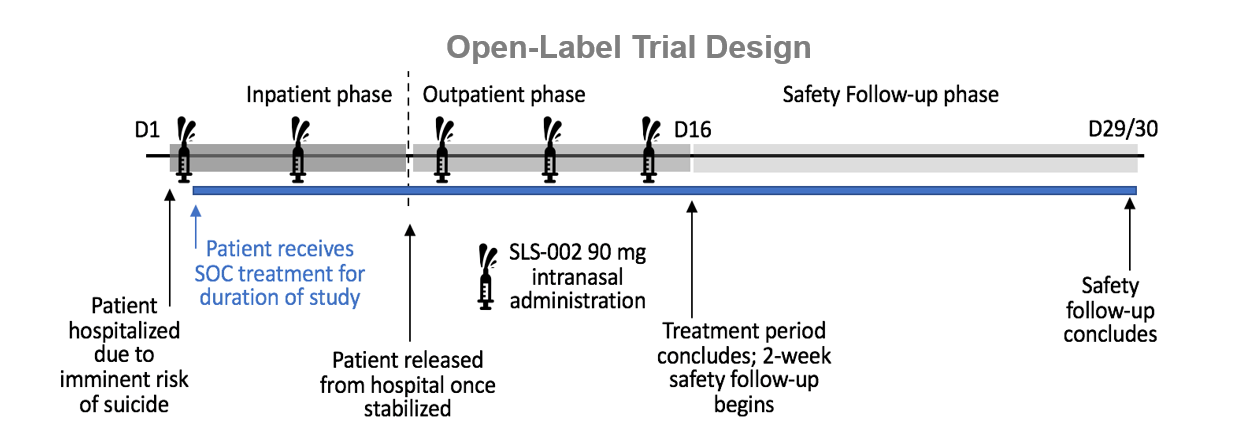
SLS-002-201 is a 2-Part Study
- Part 1 – Pilot, Open-Label – Completed (17 subjects)
- Part 2 – Double-Blind, Placebo-Controlled - Completed (147 subjects)
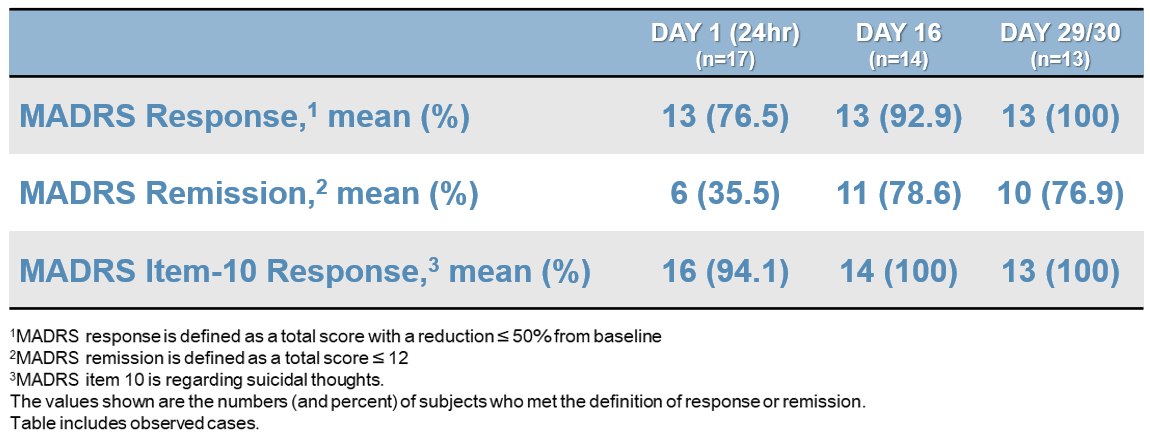
SLS-002-201 STUDY – PART 1 EFFICACY RESULTS
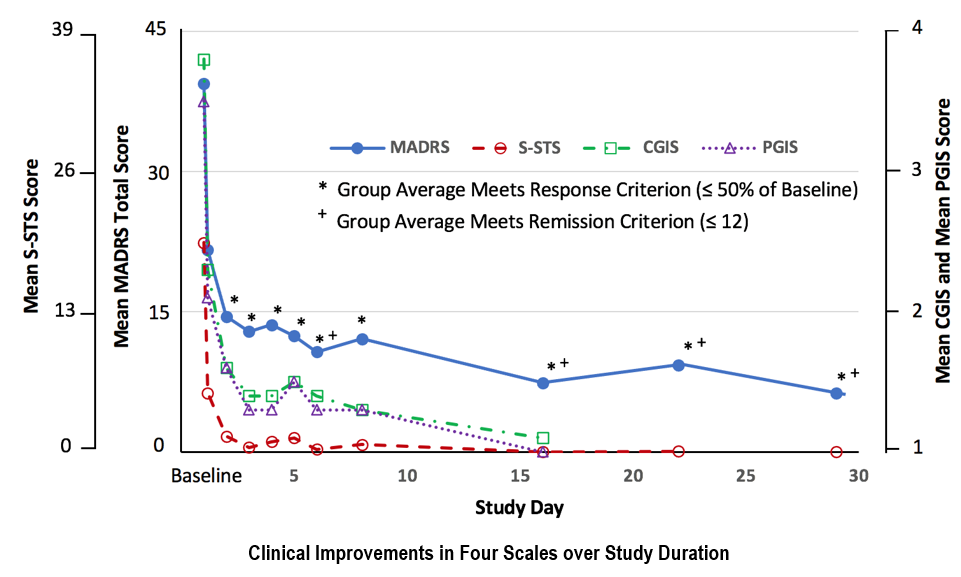
SLS-002 SHOWED CLINICAL IMPROVEMENTS ACROSS ALL FOUR SCALES
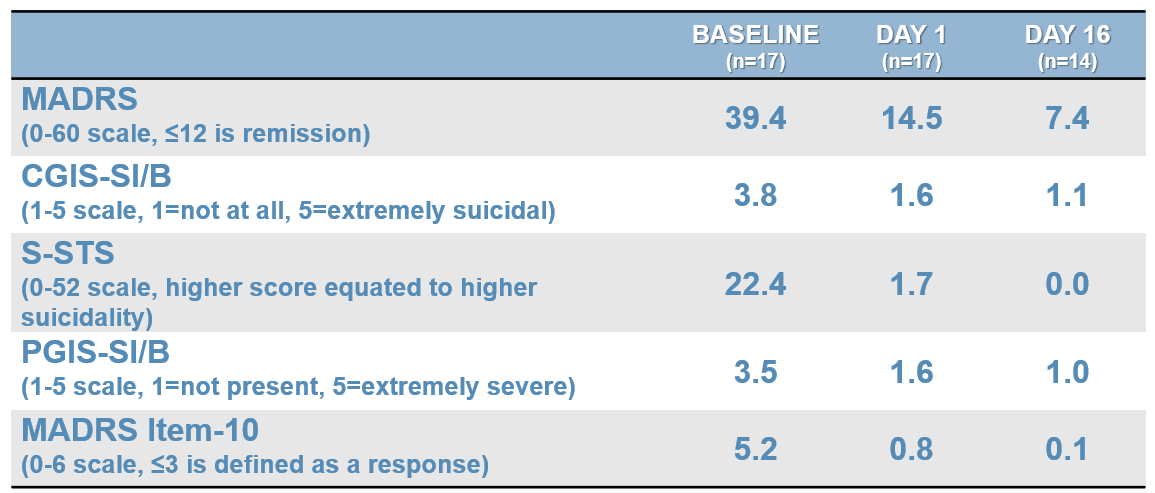
SLS-002 MADRS RESPONSE AND REMISSION
SLS-002-201 STUDY – PART 2 EFFICACY RESULTS
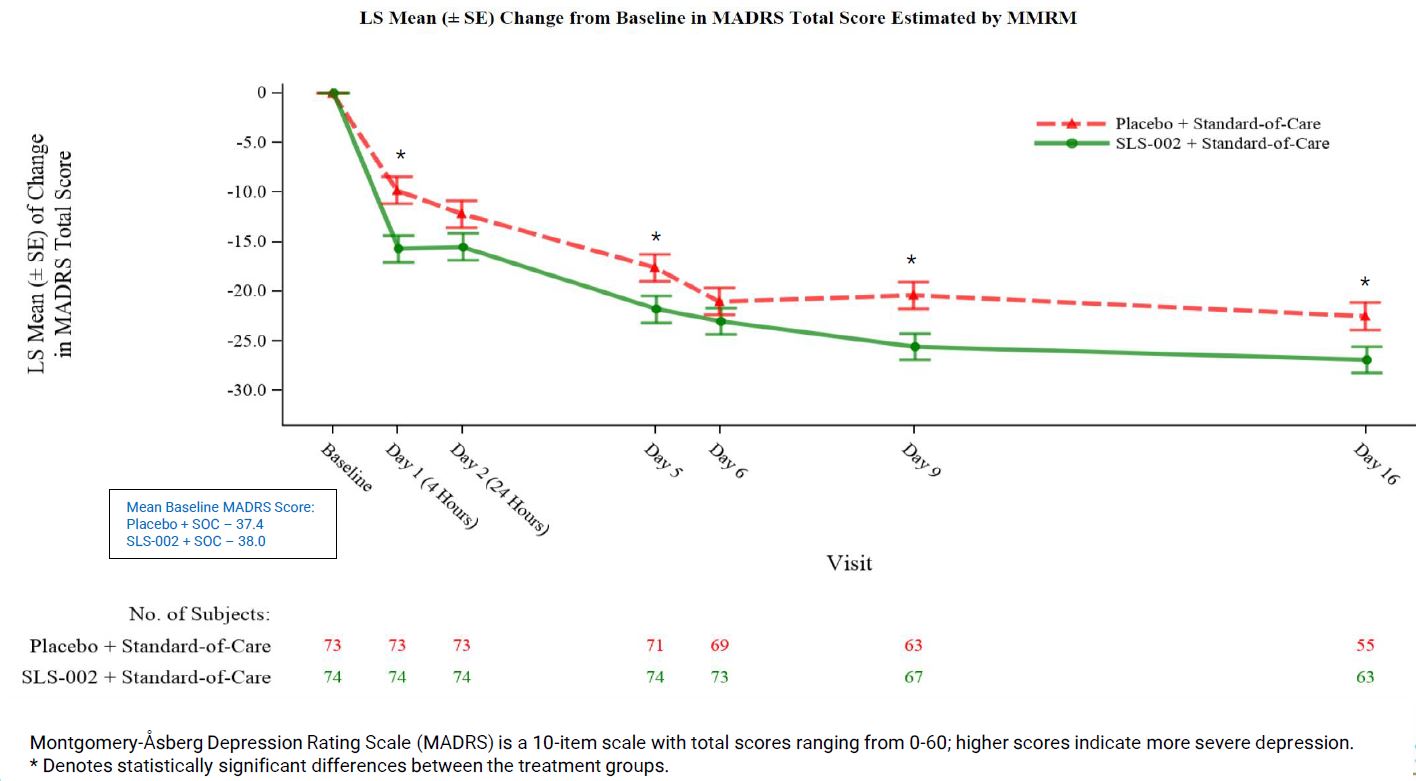
MADRS DEMONSTRATED EARLY AND CONTINUED BENEFIT THROUGHOUT THE STUDY
MADRS DEMONSTRATED CLINICALLY MEANINGFUL DIFFERENCES BETWEEN GROUPS THROUGHOUT THE STUDY
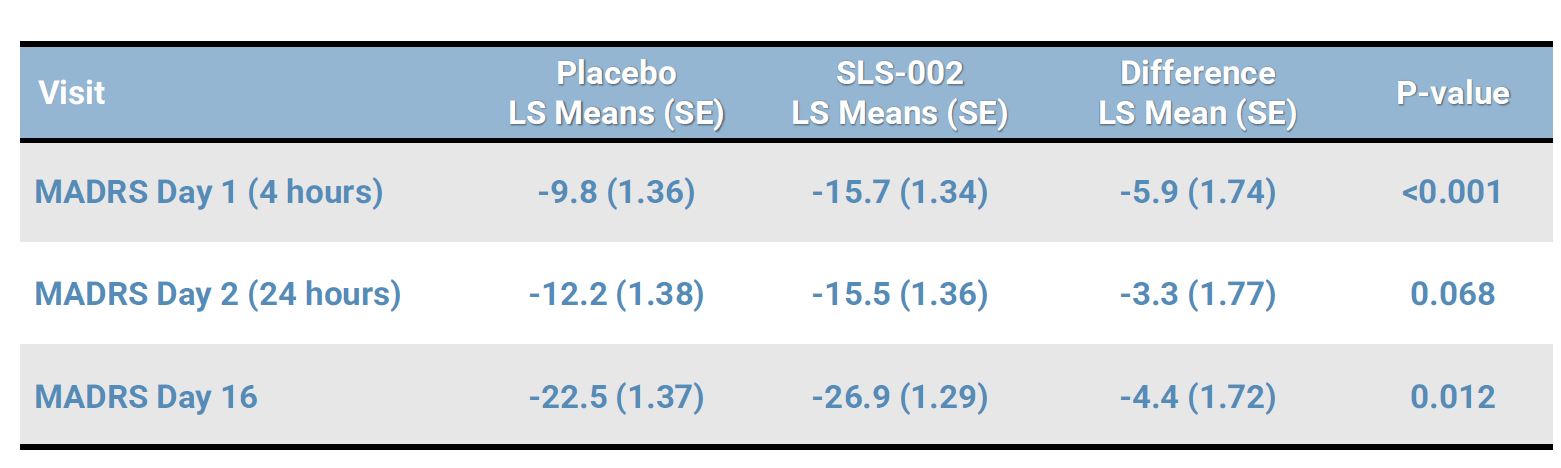
HOWEVER, STATISTICAL SIGNIFICANCE WAS NOT ACHIEVED FOR THE PREDEFINED PRIMARY ENDPOINT (ANCOVA MADRS 24 HOURS POST DOSING)
CHANGE FROM BASELINE MADRS TOTAL SCORE MMRM ESTIMATES PRESENTED HERE
SLS-002-201 SUSTAINED IMPROVEMENT ON MADRS DURING SAFETY FOLLOW-UP
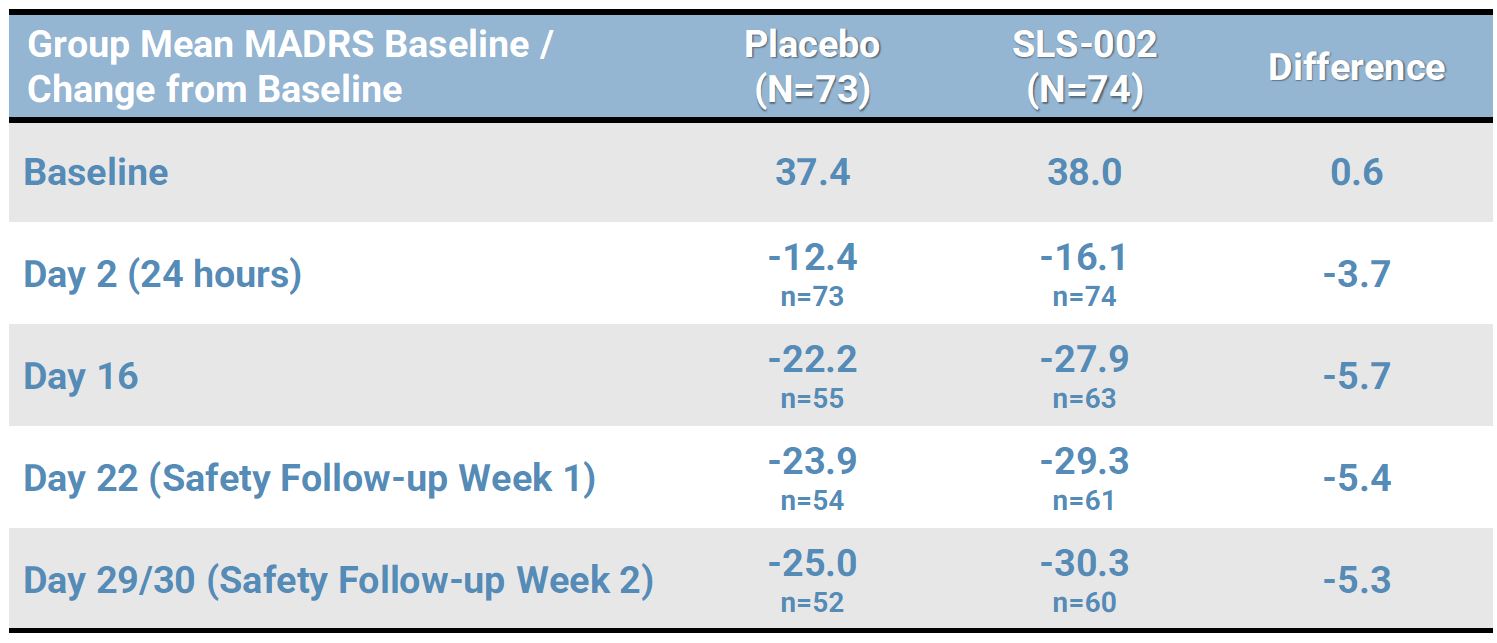
Note: Results presented are based on simple summary statistics, not adjusted (least-square) means as presented for the MMRM analyses, with differences calculated from group mean scores rounded to the tenth.
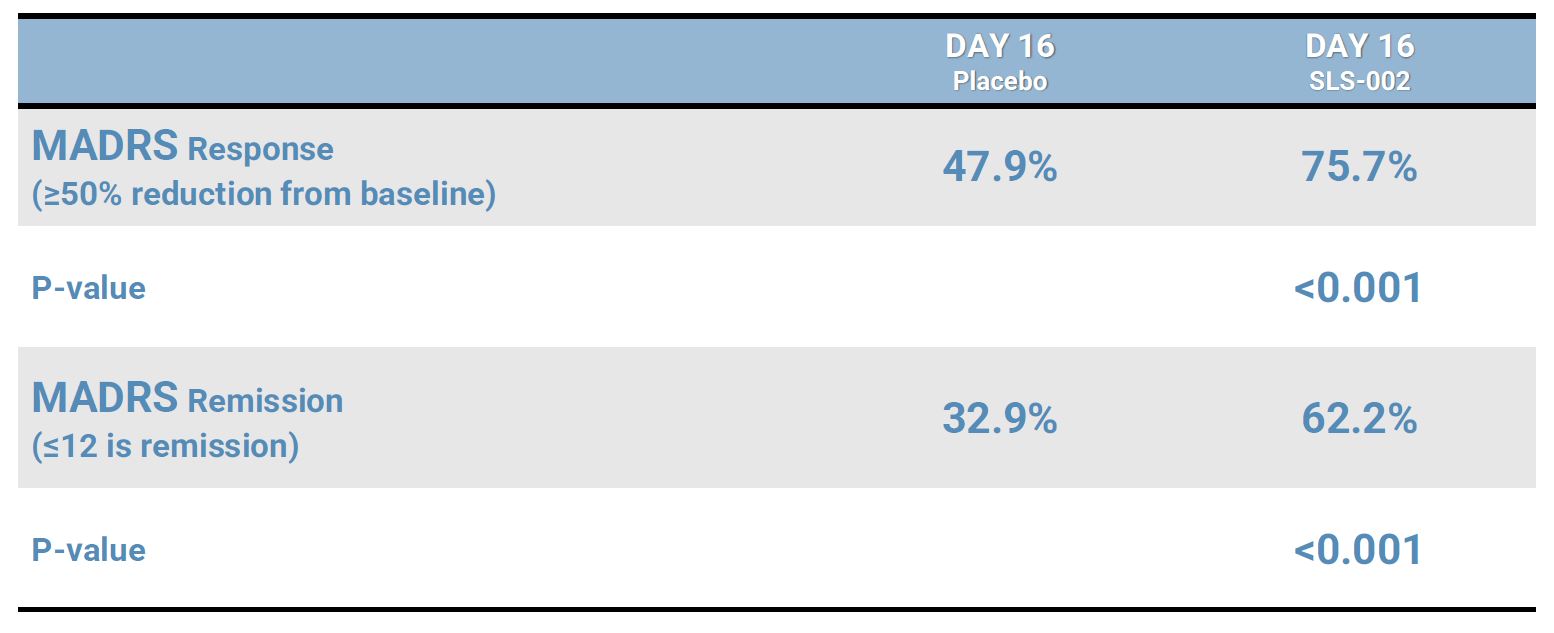
MADRS RESPONSE AND REMISSION RATES AT END OF TREATMENT
SUICIDALITY MEASURES DEMONSTRATED ACUTE DIFFERENCES BETWEEN GROUPS
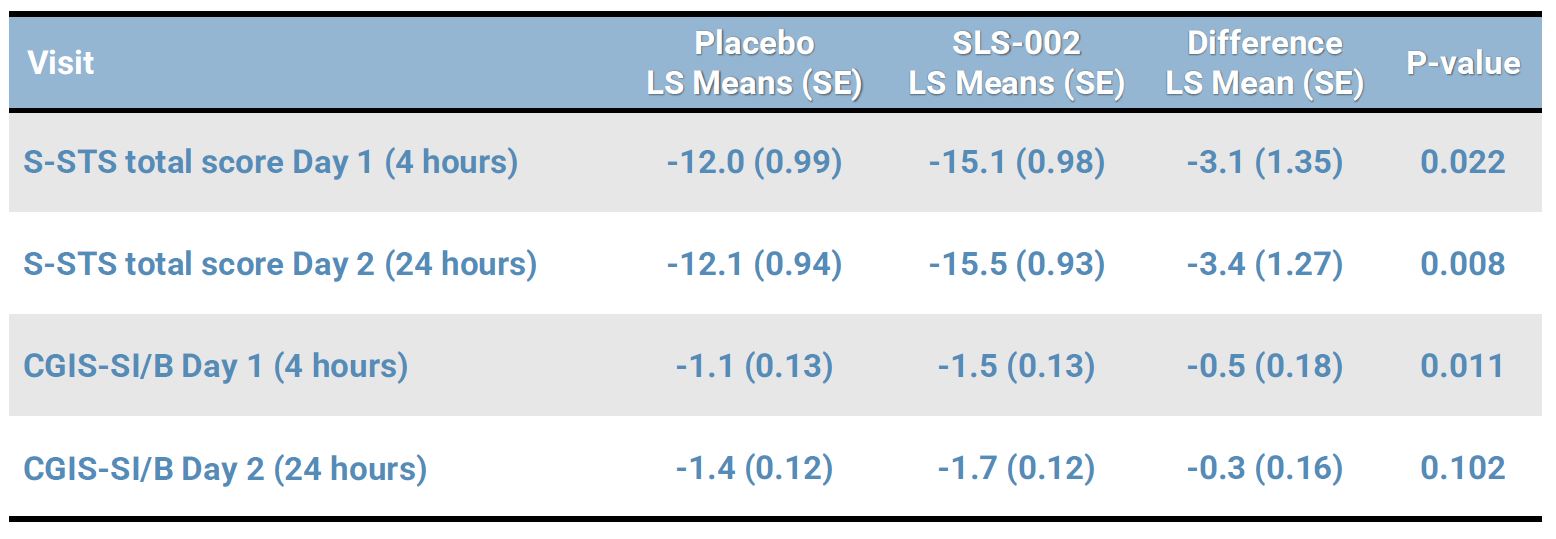
Sheehan-Suicidality Tracking Scale (S-STS) is a clinician-rated scale which includes 13 suicidality items that are rated on a scale ranging from 0 (not at all) to 4 (extremely) which yields a total score ranging from 0 to 52.
Clinical Global Impression of Severity for Suicidal Ideation and Behavior (CGIS-SI/B) is a 5-point clinician-rated measure of suicidality-specific symptom severity, ranging from 1 (not at all suicidal) to 5 (among the most extremely suicidal).
SLS-002 Intranasal Device Administration

- Manufactured by Aptar Pharma (NYSE:ATR)
- Seelos has Co-Exclusive Use of Bidose (BDS) Liquid System
- Bi-dose device administers 0.1 mL sprays in each nostril
- Disposable device for single use
- Convenient, ready to use administration