About SLS-005 (intravenous trehalose)
Trehalose is a low-molecular-weight disaccharide (0.342 kDa) that protects against pathological processes in cells.
Intravenous trehalose has been shown to penetrate muscle and cross the blood-brain barrier.
Two U.S. patents for parenteral administration of trehalose exist for patients with OPMD and SCA3; both are expected to expire in 2033. In addition, Orphan Drug Designation for OPMD, Sanfilippo syndrome and SCA3 has been secured in the U.S. and in the EU.
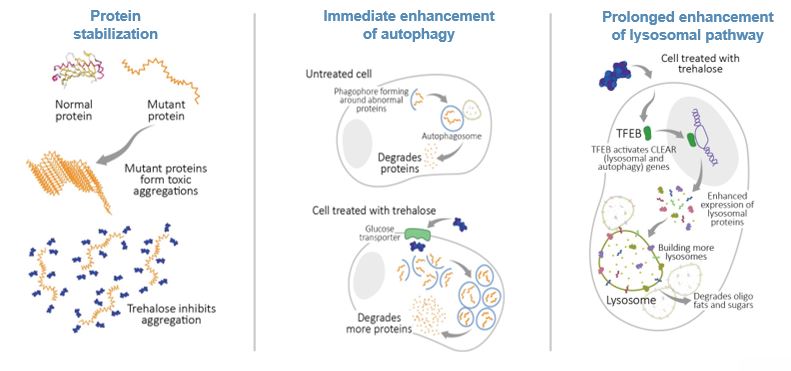
Trehalose Mechanism of Action
- Mayer AL, Higgins CB, Heitmeier MR, et al. SLC2A8 (GLUT8) is a mammalian trehalose transporter required for trehalose-induced autophagy. Scientific Reports. 6 (38586).
Available at: https://www.nature.com/articles/srep38586#f1 Accessed February 2019. - Uchida K, Unuma K, Funakoshi T, et al. Activation of Master Autophagy Regulator TFEB During Systemic LPS Administration in the Cornea. Journal Toxicol Pathol. 2014;27(2):153–8.
About Amyotrophic Lateral Sclerosis (ALS or Lou Gehrig’s disease)
- Amyotrophic lateral sclerosis (ALS) is a group of rare neurological diseases that mainly involve the nerve cells (neurons) responsible for controlling voluntary muscle movement.
- Voluntary muscles produce movements like chewing, walking, and talking.
- The disease is progressive, meaning the symptoms get worse over time.
- Currently, there is no cure for ALS and no effective treatment to halt, or reverse, the progression of the disease.
- Mutations in the C9orf72, SOD1, FUS, and TARDBP genes can cause familial ALS and contribute to the development of sporadic ALS.
- These mutations contribute to the death of motor neurons and ALS affected motor neurons develop a buildup of protein aggregates such as TDP-43 and SOD1.
SLS-005 (intravenous trehalose) for ALS
- In in-vivo studies of ALS, trehalose has been shown to increase the clearance of TDP-43, decrease SOD1 and SQSM1/p62 aggregates and monomers, delay the progression of the disease, preserve ventral horn motor neurons and increase muscle fiber size.
- Seelos completed enrollment of a Phase IIb/III trial with 160 patients with either familial or sporadic ALS in a double-blind placebo-controlled trial.
- Patients were randomized 3:1 (drug:placebo) and studied with a primary endpoint measuring the change from baseline on Revised Amyotrophic Lateral Sclerosis Functional Rating Scale (ALSFRS-R) score at 24 weeks. Secondary endpoints were measured at 24 weeks, including the change from baseline in slow vital capacity, muscle strength, quality of life measurements as well as additional signs of disease progression.
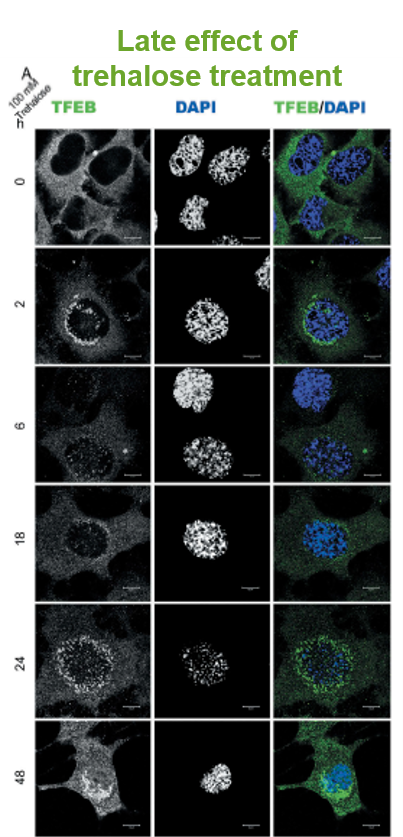
Induces autophagy via lysosomal-mediated TFEB activation in models of motoneuron degeneration
- Impairment of TFEB has been demonstrated as a key element in the pathogenesis of ALS
- Immortalized motor neuron cell line (NSC34)
- Trehalose exposure
- TFEB translocated to the nucleus
- Activates genes for autophagy
- Increased clearance of TDP-43
- Rusmini P, Cortese K, Crippa V, et al. Trehalose induces autophagy via lysosomal-mediated TFEB activation in models of motoneuron degeneration. Autophagy. 2019;15(4):631-651. doi:10.1080/15548627.2018.1535292
- Wang Y, Liu FT, Wang YX, et al. Autophagic Modulation by Trehalose Reduces Accumulation of TDP-43 in a Cell Model of Amyotrophic Lateral Sclerosis via TFEB Activation. Neurotox Res. 2018;34(1):109-120. doi:10.1007/s12640-018-9865-7
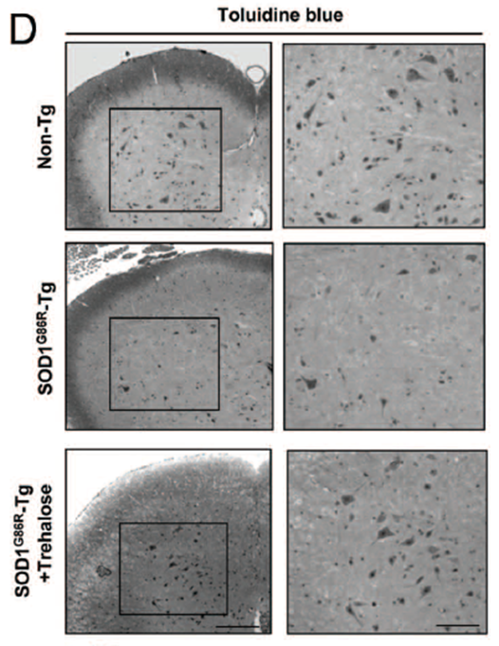
Trehalose delays the progression of ALS by enhancing autophagy in motoneurons
- Intraperitoneal injection (or IP injection) and oral trehalose (combined)
- Improves survival of male>female
- Delays progression of the disease
- Decreased SOD1 aggregates and monomers
- Preserved ventral horn motor neurons
- Induced autophagy
- Castillo K, Nassif M, Valenzuela V, et al. Trehalose delays the progression of amyotrophic lateral sclerosis by enhancing autophagy in motoneurons. Autophagy. 2013;9(9):1308-1320. doi:10.4161/auto.25188
SLS-005 (intravenous trehalose) for SCA3
Intravenous trehalose, which is currently an investigational molecule, has demonstrated favorable safety and efficacy in 59 patients across two Phase II studies in rare diseases (OPMD and SCA3).
- SCA3 (or Machado-Joseph disease) is an autosomal dominant cerebellar ataxia (ADCA I) due to CAG repeat expansions in ataxin
- Adult-onset neurological PolyQ disease that begins between the ages of 20 and 50
- Orphan designation (US/EU)
- Most commonly affects individuals of Portuguese/ Azorean descent
- Coutinho P, Andrade C. Autosomal dominant system degeneration in Portuguese families of the Azores Islands: a new genetic disorder involving cerebellar, pyramidal, extrapyramidal and spinal cord motor functions. Neurology. 1978;28 (7) 703- 709